1 Awassa College of Agriculture, P.O. Box 5, Awassa, Ethiopia
2 E (Kika) de la Garza Institute for Goat Research, Langston University, Langston, OK, USA
Introduction
There are currently 5 million goats in the USA, with potential to produce mohair, meat, and cashmere worth $550 million/year. Recently, interest in goat meat has risen, fueled by the increasing demand by ethnic populations within the USA. Despite the spiraling number of goats slaughtered in the USA, the demand for goat meat is much greater than the domestic supply. Thus, considerable goat meat is imported from Australia and New Zealand. In 1990, the USA imported 1,362 metric tons of chilled or frozen goat meat, at a value of $1.86 million. By 1992, imports rose by 70% to 2,362 metric tons, with a value of $3.6 million.
The domestic shortfall in goat production can be addressed by either increasing the number of goats or improving production of the existing population (Poore, 1993). In regard to the latter mode, internal and external parasites seriously limit both per head productivity and the density at which goats can be raised on farms. In ruminants, high internal nematode burdens suppress live weight gain, reproductive efficiency, milk and wool production (Blackburn et al., 1991) and sometimes even result in death (Rahman and Collins, 1990). In the Australian and New Zealand sheep industries, the annual economic loss attributed to nematodes is estimated at $400 (Gray, 1987) and $270 million (Parker, 1991), respectively, despite an anthelmintic expenditure of $50 million by Australian farmers (Gray, 1987). Similar estimates are not readily available for the USA small ruminant industry, but it is believed that economic losses are of similar magnitude (SR-CRSP, 1991).
Conventional methods to control internal parasites include anthelmintic drenching, pasture spelling, crop rotation, and integrated grazing systems using stock with low internal parasite susceptibility. Anthelmintic use, by far the least complex of control options, is currently the preferred method for internal parasite management (Jackson, 1991; Zajac and Moore, 1993). Unfortunately, after many years of heavy reliance on anthelmintics for internal parasite control, parasite resistance has emerged. Anthelmintic resistance has been documented across the world, including Africa (Maingi, 1991), Asia (Dorny et al., 1993), and North America (DeVaney et al., 1992). Anthelmintic resistance of internal parasite populations exists for all current drug families including benzimidazole (McKenna, 1990), levamisole (Verady et al., 1993), ivermectin (DeVaney et al., 1992), and tomorantel (Anderson et al., 1988). In some studies, internal parasite populations have multiple resistances. To avoid anthelmintic resistance, goat producers must reduce reliance on anthelmintics and seek alternative, sustainable methods for internal nematode control.
Medicinal plants such as Hagenia abyssinica (Kosso) and Glinus lotoides (Hirta) contain substances with nematocidal activity (Abegaz and Dagne, 1978). The ability to control nematode infestation in humans through use of plants with anthelmintic properties has been recognized and is widely used in countries such as Ethiopia (Abegaz and Dagne, 1978; Sofowora, 1982). However, potential use of these plants as anthelmintics and optimal dosing regimes for ruminants have yet to be identified. Thus, the objective of this research was to examine the anthelmintic activity of Hagenia abyssinica (Kosso) in growing Alpine goats.
Material and Methods
Fresh fecal samples were collected from Alpine goats not previously drenched with anthelmintics for one month prior to the experiment. Goats with fecal egg count burdens in excess of 1,000 eggs/gram of feces (for a total of at least 42 goats) had three consecutive daily pretreatment fecal samples collected for egg concentration determination using a McMaster chamber. Fecal eggs were categorized by type. Goats were blocked according to worm infestation, and plant extracts were administered once orally. Control goats were administered solvent or basal solution, and the three nonzero dose levels were 20, 40 and 60 g dry matter of Kosso. Fecal egg count were taken at 24, 48, 72, and 96 hours postdosing to determine the reduction in fecal egg counts. Count data was transformed using log(count+10) and packed cell volume was transformed using arcsin function. Repeated measures were analyzed using PROC MIXED of SAS. Otherwise, PROC GLM was used.
Results and Discussion
Heterogeneity of slope was similar among variables except for the number of trichuris eggs (Table 1), indicating that for all variables except trichuris eggs treatment effects were similar among the different days of sampling. The only variable affected by oral dosing with Kosso was the number of cestode eggs, being decreased. The appearance of cestodes in feces occurred soon after dosing, starting only 1 day later. Different levels of Kosso were given to discern a minimal level of Kosso necessary for anthelmintic effects. However, the results obtained suggest need for further study of Kosso levels, since cestode egg levels were similar among nonzero levels and numerically the number was greater for the 60 vs 20 and 40 g doses.
Conclusions
Kosso appears to hold promise as an anthelmintic for cestode control in goats. However, further research is needed to determine optimal levels of Kosso, as well as to identify mode of action and specific fraction(s) of Kosso with anthelmintic activity. Furthermore, there should be experimentation with other plants possibly possessing anthelmintic activities, such as Glinus lotoides (Hirta). Such work might one day yield low-cost and sustainable methods of internal parasite control for use in goat production systems.
Acknowledgement
This research was supported by USDA/FAS/ICD/RSED Project Number 58-3148-0914.
References
Abegaz, B, and Dagne, E. 1978. Comparative bioassay studies of some traditional anthelmintic plants, plant extracts and modern drugs. Sinet: Ethiopia J. Sci. 1:117-121.
Anderson, N., Martin, P.J., and Jarrett, R.G. 1988. Mixtures of anthelmintics: A strategy against resistance. Aust. Vet. J. 65(2):62-63.
Blackburn, H.D., Rocha, J.L., Figueiredo, E.P., Berne, M.E., Vieira, L.S., Cavalcante, A.R., and Rosa, J.S. 1991. Interaction of parasitism and nutrition and their effects on production and clinical parameters in goats. Vet. Parasitol. 40:99-112.
DeVaney, J.A., Craig, T.M., and Row, L.D. 1992. Resistance to ivermectin by Haemonchus contortus in goats and calves. Int. J. Parasitol. 22(3):369-376.
Dorny, P., Claerebout, E., Vercruysse, J., Jaila, A., and Sani, R. 1993. Benzimidazole resistance of Haemonchus contortus in goats in Malaysia. Vet. Record 133:423-424.
Gray, G.D. 1987. Genetic resistance to haemonchosis in sheep. Parasitol. Today 3(8):253-255.
Jackson, F. 1991. Anthelmintic resistance in goats. Goat Vet. Soc. J. 12(1):1-6.
Maingi, N. 1991. Resistance to thiabendazole, fenbendazole and levamisole in Haemonchus and Trichostrongylus species in sheep on a Kenyan farm. Vet. Parasitol. 39:285-291.
McKenna, P.B. 1990. The detection of anthelmintic resistance by faecal egg count reduction test: An examination of some of the factors affecting performance and interpretation of. N.Z. Vet. J. 38:142-147.
Parker, A.G.H. 1991. Selection for resistance to parasites in sheep. Proc. N.Z. Soc. Anim. Sci. 51:291-294.
Poore, M.H. 1993. Important factors to consider in meat goat management. Meat Goat Field Day Handout, October 9, 1993, North Carolina State University, Raleigh, NC.
Rahman, W.R., and Collins, G.H. 1990. Changes in liveweight gain, blood constituents and worm egg output in goats artificially infected with a sheep-derived strain of Haemonchus contortus. Br. Vet. J. 146:543-550.
SR-CSRP. Breeding a genetically improved dual purpose goat adapted for production in Kenya. Small Ruminant – Collaborative Research Support Program 1990-91 Annual Report, USAID. pp 9-23. University of California, Davis, Davis, CA.
Sofowora, A. 1982. Medicinal plants and traditional medicine in Africa. pp.75, John Wiley and Sons limited.
Verady, M., Praslicka, J., Corbra, J., and Vesley, L. 1993. Multiple anthelmintic resistance of nematodes in imported goats. Vet. Rec. 132(15):387-388.
Zajac, A.M. and Moore, G.A. 1993. Treatment and control of gastrointestinal nematodes in sheep. Compend. Cont. Educ. Prac. Vet. 15(7):999-1011.
Table 1. Effects of level of oral administration of Hagenia abyssinica (Kosso) on measures of internal parasitism in growing Alpine goats
|
Treatment |
||||
|
0 |
20 |
40 |
60 |
|
|
Beginning weight, kg |
16.3a |
17.7a |
20.6a |
17.0a |
|
Ending weight, kg |
18.9a |
20.4a |
22.7a |
21.1a |
|
Packed cell volume, % |
20.8a |
23.3a |
22.9a |
21.6a |
|
Egg counts, egg/g feces |
||||
|
Cestodes |
1372.3a |
171.5b |
163.3b |
707.2b |
|
Eimeria |
984.6a |
854.4a |
1459.5a |
607.4a |
|
Strongyles |
1877.3a |
1589.2a |
1840.3a |
1897.9a |
|
Trichuris |
1.4a |
4.0a |
5.7a |
0.3a |
|
Heterogeneity of slope for counts, egg/gr |
||||
|
Cestodes |
22.454a |
-6.423b |
-6.653b |
-5.114b |
|
Eimeria |
10.144a |
6.404a |
6.599a |
4.381a |
|
Strongyles |
-7.709a |
-1.242a |
-10.291a |
-20.041a |
|
Trichuris |
-0.006a |
0.414a |
1.178b |
-0.037a |
|
Heterogeneity of slope for packed cell |
0.014a |
0.020a |
0.026a |
0.054a |
Figure 1. Eggs per gram of feces for cestodes.
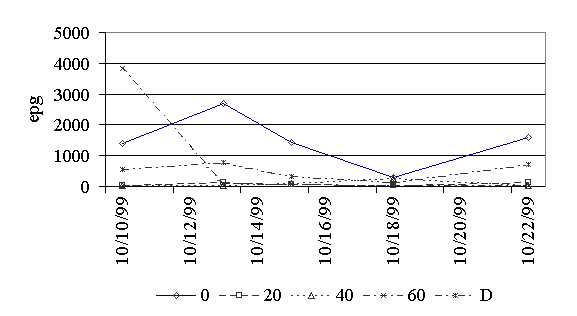
Figure 2. Eggs per gram of feces for strongyles.
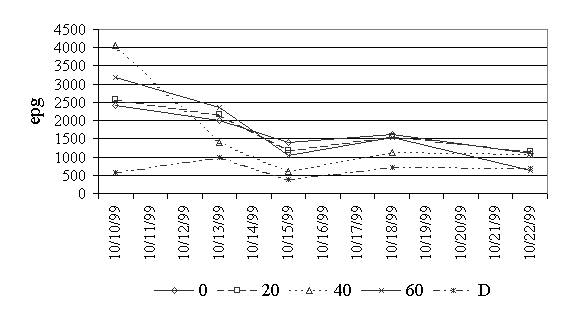
Figure 3. Eggs per gram of feces for trichuris.
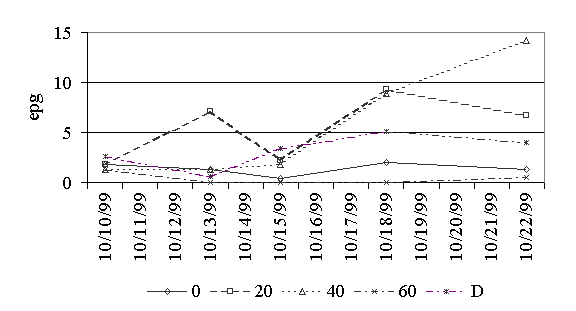
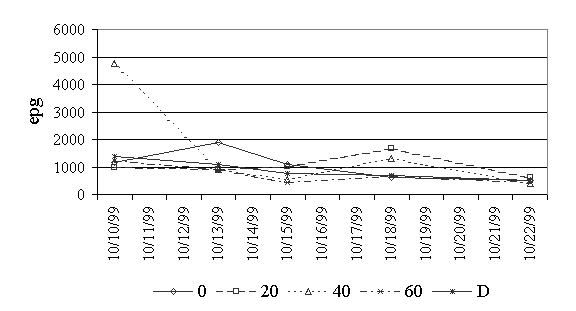
Figure 4. Oocystes per gram of feces for eimeria.
Citation:
Abebe, G., L.J. Dawson, G. Detweiler, T.A. Gipson and T. Sahlu. 2000. Hagenia abyssinica (Kosso) for internal parasite control in goats. In: R.C. Merkel, G. Abebe and A.L. Goetsch (eds.). The Opportunities and Challenges of Enhancing Goat Production in East Africa. Proceedings of a conference held at Debub University, Awassa, Ethiopia from November 10 to 12, 2000. E (Kika) de la Garza Institute for Goat Research, Langston University, Langston, OK pp. 190-195.
